Bleach
Scientific name: Sodium Hypochlorite

The main chemical compound forming bleach is sodium hypochlorite (also known as chlorine gas), which is then mixed with water. Therefore the chemical formula would be: NaCIOH2O (CIO or hyplochlorite is an ion.)
A few common names associated with Bleach are Clorox, Rinser, Hydrogen Peroxide, Chlorine, and Disinfectant, just to name a few.
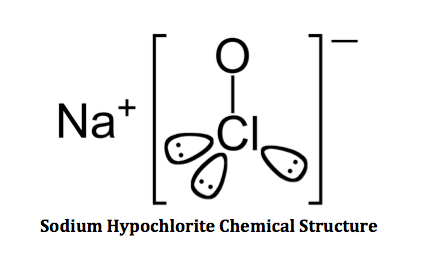
Sodium hypochlorite is composed of one sodium (Na) atom, one chlorine (Cl) atom and one oxygen (O) atom. The chemical structure shows that the hypochlorite ion (ClO-) has an ionic bond with the sodium (Na+) ion. The H20 used to create bleach is composed of 2 hydrogen atoms and one oxygen atom.
Physically, sodium hypochlorite is a clear liquid. Mixed with H20 to form bleach, it becomes a thick, white liquid. It has the odor of chlorine. Chemically, it is a strong oxidizer and corrosive. It burns the skin and irritates eyes. Mixing something such as an acid cleaner with bleach, can cause a toxic reaction that forms chlorine gas. It also reacts with acetic acid to produce hypochlorous acid. It decomposes when heated, forming sodium chlorate and sodium chloride. Dilutions above 550 ppm are corrosive to metals and plastics, but some bleaching agents are diluted this high. Bleach is normally 1 part out of 9 times water for safety reasons.
A few common names associated with Bleach are Clorox, Rinser, Hydrogen Peroxide, Chlorine, and Disinfectant, just to name a few.
Sodium hypochlorite is composed of one sodium (Na) atom, one chlorine (Cl) atom and one oxygen (O) atom. The chemical structure shows that the hypochlorite ion (ClO-) has an ionic bond with the sodium (Na+) ion. The H20 used to create bleach is composed of 2 hydrogen atoms and one oxygen atom.
Physically, sodium hypochlorite is a clear liquid. Mixed with H20 to form bleach, it becomes a thick, white liquid. It has the odor of chlorine. Chemically, it is a strong oxidizer and corrosive. It burns the skin and irritates eyes. Mixing something such as an acid cleaner with bleach, can cause a toxic reaction that forms chlorine gas. It also reacts with acetic acid to produce hypochlorous acid. It decomposes when heated, forming sodium chlorate and sodium chloride. Dilutions above 550 ppm are corrosive to metals and plastics, but some bleaching agents are diluted this high. Bleach is normally 1 part out of 9 times water for safety reasons.